|
3/10/2024 0 Comments 757 mmhg to atm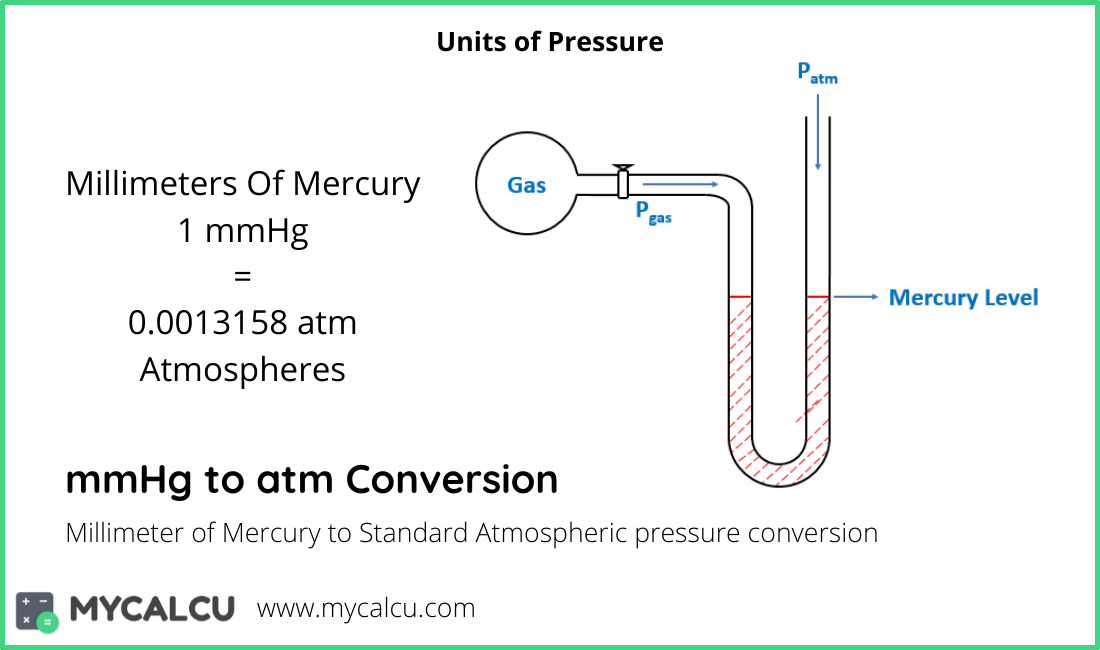 
Type in unit symbols, abbreviations, or full names for units of length, area, mass, pressure, and other types. Task: Convert 975 mmHg to atmospheres (show work) Formula: mmHg 760 atm Calculations: 975 mmHg 760 1.28289474 atm Result: 975 mmHg is equal to 1.28289474 atm Conversion Table For quick reference purposes, below is a conversion table that you can use to convert from mmHg to atm. 
1 psi 6,894.757 Pa: millimeters of mercury: mmHg: Non-SI (International) 1 mmHg 133.3224 Pa: bar. (Torr to mmHg) Torr to Pascal (Torr to Pa) Torr to Pounds per square. You can find metric conversion tables for SI units, as well as English units, currency, and other data. 757 mmHg, 112.1C : 1.1818 at 20/4C fl p: 5.3C (isomeric mixture) lel: 5.3 (isomeric mixture) uel: 14.5 (isomeric mixture) KH: 3.55 × 103 atm. A torr is equal to a pressure of 133.3224 pascals or 0.00131579 atmospheres. We assume you are converting between millimeter of mercury 0 ☌ and inch of mercury 0 ☌.You can view more details on each measurement unit: mmHg or inHg The SI derived unit for pressure is the pascal. How many mmHg in 1 inHg The answer is 25.399999704976. It is approximately equal to Earth's atmospheric pressure at sea level.Ĭ provides an online conversion calculator for all types of measurement units. More information from the unit converter. It is sometimes used as a reference pressure or standard pressure. Pascal : The pascal (symbol: Pa) is the SI unit of pressure which derived from other SI units. Explanation: The density of ammonia gas, NH3, at 31☌ and 751 mmHg can be calculated using the ideal gas law equation. The standard atmosphere (symbol: atm) is a unit of pressure defined as 101325 Pa (1.01325 bar). It is widely used, and its value is approximately equal to a pressure of 1 torr. Final answer: The density of ammonia gas, NH3, at 31☌ and 751 mmHg is approximately 0.785 g/L. The unit is named after Evangelista Torricelli, Italian physicist and mathematician, for his discovery of the principle of the barometer in 1643. It is the atmospheric pressure that supports a column of mercury 1 millimetre high. To convert it to atm using the calculator, you would follow these steps: Enter 760 in the input field. The torr (symbol: Torr) or millimetre of mercury (mmHg) is a non-SI unit of pressure. To convert pressure from mmHg to atm, you can use the following formula: atmmmHg760.0atm760.0mmHg Example: Suppose you have a pressure value of 760 mmHg. The barometric pressure pushing down on the liquid in the glass then forces the liquid up the straw.You can do the reverse unit conversion fromĪtmosphere to mmHg, or enter any two units below: Enter two units to convert From: It is defined as the ratio of 760 to 1standard atmosphere, approximately equals one millimeter of mercury. Visit 757.2 Atmospheres to mmHg Conversion Millimeter Mercury (0☌) : Millimeter of mercury is a small pressure unit which represents the pressure pushing down due to gravity of any volume of liquid mercury which is 1mm high. Torr : The torr (the symbol: Torr) is a unit of measurement for pressure, but it is a non-SI unit. We can use a drinking straw because sucking on it removes air and thereby reduces the pressure inside the straw. It is widely used, and its value is approximately equal to a pressure of 1 torr. We make use of barometric pressure in many ways. 0.99342 Atmospheres (atm) Visit 755 Atmospheres to mmHg Conversion Millimeter Mercury (0☌) : Millimeter of mercury is a small pressure unit which represents the pressure pushing down due to gravity of any volume of liquid mercury which is 1mm high. It is released and rises to an altitude of 8. A helium-filled weather balloon has a volume of 616 L at 22.9☌ and 757 mmHg. Instead, what we notice are changes in the pressure, such as when our ears pop in fast elevators in skyscrapers or in airplanes during rapid changes in altitude. P1 752 mmHg / 760 0.989 atm P2 522 mmHg / 760 0.687 atm Substitute known values into the combined gas law and solve for V2: (0.989 atm × 811 L) / 293.05 K (0.687 atm × V2) / 266.05 K. 
We are so accustomed to living under this pressure that we never notice it. Task: Convert 325 mmHg to kilopascals (show work) Formula: mmHg x 0.1333223684 kPa Calculations: 325 mmHg x 0.1333223684 43.32977 kPa Result: 325 mmHg is equal to 43.32977 kPa. Thus a pressure of 1 atm equals 760 mmHg exactly.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
